J. Guo, P. M. Tolstoy, B. Koeppe, N. S. Golubev, G. S. Denisov, S. N. Smirnov, H.-H. Limbach
«Hydrogen Bond Geometries and Proton Tautomerism of Homo-Conjugated Anions of Carboxylic Acids Studied via H/D Isotope Effects on 13C NMR Chemical Shifts»
J. Phys. Chem. A 2012, ASAP.
DOI: 10.1021/jp304943h.

Abstract:
Ten formally symmetric anionic OHO hydrogen bonded complexes, modeling Asp/Glu amino acid side chain interactions in nonaqueous environment (CDF3/CDF2Cl solution, 200–110 K) have been studied by 1H, 2H, and 13C NMR spectroscopy, i.e. intermolecularly H-bonded homoconjugated anions of acetic, chloroacetic, dichloroacetic, trifluoroacetic, trimethylacetic, and isobutyric acids, and intramolecularly H-bonded hydrogen succinate, hydrogen rac-dimethylsuccinate, hydrogen maleate, and hydrogen phthalate. In particular, primary H/D isotope effects on the hydrogen bond proton signals as well as secondary H/D isotope effects on the 13C signals of the carboxylic groups are reported and analyzed. We demonstrate that in most of the studied systems there is a degenerate proton tautomerism between O-H···O– and O–···H–O structures which is fast in the NMR time scale. The stronger is the proton donating ability of the acid, the shorter and more symmetric are the H-bonds in each tautomer of the homoconjugate. For the maleate and phthalate anions exhibiting intramolecular hydrogen bonds, evidence for symmetric single well potentials is obtained. We propose a correlation between H/D isotope effects on carboxylic carbon chemical shifts and the proton transfer coordinate, q1 =1/2(rOH – rHO), which allows us to estimate the desired OHO hydrogen bond geometries from the observed 13C NMR parameters, taking into account the degenerate proton tautomerism.
Завершена установка просвечивающих электронных микроскопов Jeol JEM-1400 и Jeol JEM-2100. С 17го июля на микроскопы будут устанавливаться цифровые камеры. Завершается установка сканирующего электронного микроскопа Tescan MIRA3 LMU. Начинается установка газового хромато-масс-спектрометра LECO Pegasus 4D.
На спектрометре Bruker DPX 300 для пользователей из СПбГУ в июне было снято всего 243 ЯМР спектра на ядрах 1H и 13C.
Gurinov, A. A.; Mauder, D.; Akcakayiran, D.; Findenegg, G. H.; Shenderovich, I. G.
«Does Water Affect the Acidity of Surfaces? The Proton-Donating Ability of Silanol and Carboxylic Acid Groups at Mesoporous Silica.»
ChemPhysChem 2012, 13 (9), 2282-2285.
DOI: 10.1002/cphc.201200204
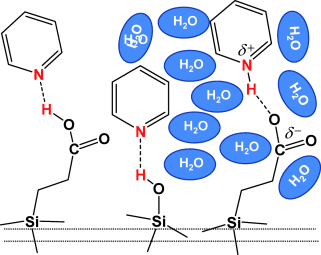
Solvation at the interphase:
A study of the influence of water on the effective acidity of silanol and carboxylic acid groups of propionic acid functionalized SBA-15 reveals that to affect the proton-donating ability of an acidic group at the surface, water should be able to form a solvation shell around that group. As a result, water does not affect the acidity of native SBA-15 but dramatically enhances that of SBA-15 functionalized with propionic acid moieties.
Начинается установка оборудования для просвечивающей электронной микроскопии. Устанавливаются ПЭМ Jeol JEM-1400 STEM, ПЭМ Jeol JEM-2100 HC, Вакуумная напылительная станция Jeol JEE-420D , Система документации DitaBis Vario, Плазменная очистка держателя и образца Jeol JIC-410 , Система обезгаживания плёнки JEOL Photoplate degasser , Вакуумная станция Gatan 655 ,Криодержатель томографический Gatan 914, Настольный СЭМ Jeol JCM-5000 Neoscope.
Установка и настройка оборудования ориентировочно продлится 4 недели, до 12 июля 2012 года.
Подробнее.




