Mikhail A. Kinzhalov, Konstantin V. Luzyanin, Irina A. Boyarskaya, Galina L. Starova, Vadim P. Boyarskiy
«Synthetic and structural investigation of [PdBr2(CNR)2] (R = Cy, Xyl)»
Journal of Molecular Structure 2014, 1068, 222–227
DOI: 10.1016/j.molstruc.2014.04.025
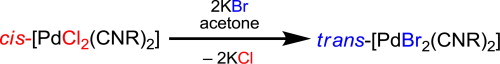
Reaction of cis-dichloro-bis-(cyclohexylisocyanide)palladium(II) ([PdCl2(CNCy)2], (1) or cis-dichloro-bis-(2,6-dimethylphenyl-isocyanide)palladium(II) ([PdCl2(CNXyl)2], (2) with excess of potassium bromide in acetone furnished complexes trans-dibromo-bis-(cyclohexylisocyanide)palladium(II) ([PdBr2(CNCy)2], (3) or trans-dibromo-bis-(2,6-dimethylphenylisocyanide)palladium(II) ([PdBr2(CNXyl)2], (4), respectively. Both compounds were characterized by elemental analyses (C, H, N), high resolution ESI+-MS, IR, 1H and 13C{1H} NMR spectroscopies, and their crystals were analyzed by a single-crystal and powder X-ray diffraction methods. To rationalise the dependence of the structure of 1–4 on type of halogen ligands, the Gibbs free energies of corresponding cis- and trans-isomers of 1–4 were estimated at the DFT B3LYP level of theory.
Д.ф.-м.н. Константин Иванов из “Международного томографического центра СО РАН” прочитал лекцию «Методы спиновой гиперполяризации в ЯМР».
27 мая в 15:00 на химическом факультете в помещении 1093 (конференц-зал РЦ РДМИ) состоится лекция «Методы спиновой гиперполяризации в ЯМР», которую прочтет д.ф.-м.н. Иванов Константин Львович из “Международного томографического центра СО РАН”, Новосибирск. Приглашаются все желающие.
Ludmila L. Rodina, Jury J. Medvedev, Olesia S. Galkina and Valerij A. Nikolaev
«Thermolysis of 4-Diazotetrahydrofuran-3-ones: Total Change of Reaction Course Compared to Photolysis»
European Journal of Organic Chemistry 2014, 14, 2993–3000
DOI: 10.1002/ejoc.201400161

Thermolysis of 2,2,5,5-tetrasubstituted 4-diazodihydrofuran-3-ones in protic (BnOH) and aprotic (DMSO) media, in contrast to photolysis, gives rise to the formation of 2,2,4,5-substituted 3(2H)-furanones as a result of 1,2-alkyl (aryl) shift. The thermal stability of diazodihydrofuranones in these processes is determined by the nature of the substituents at the C-5 atom of the heterocyclic structure: alkyl and electron-donating groups in the para-position of the aryl substituent notably reduce the persistence of diazodihydrofuranones, whereas aryl groups and electron-withdrawing substituents increase their stability considerably. Thermolysis of substituted 4-diazodihydrofuran-3-ones in DMSO solution is a first-order reaction that furnishes higher yields of furanones than thermolysis in BnOH. The reaction can serve as a preparative method for the synthesis of tetrasubstituted-3(2H)-furanones.
Сегодня РЦ посетил почетный директор по науке Исследовательского центра лазера на свободных электронах XFEL, Германия, проф. Йохан Шнайдер.







